
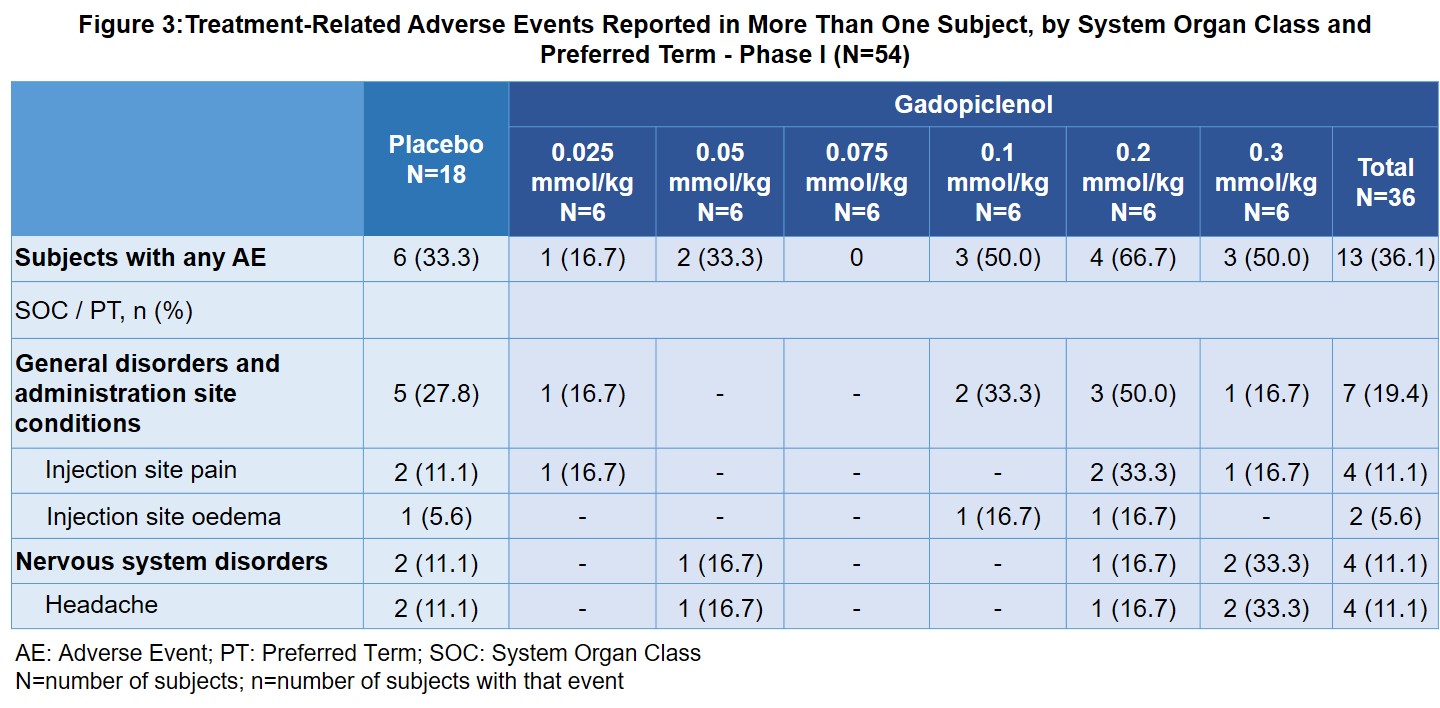
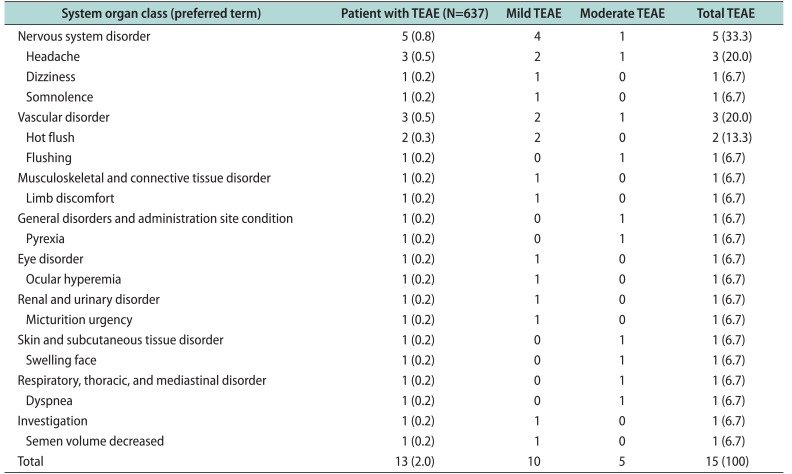
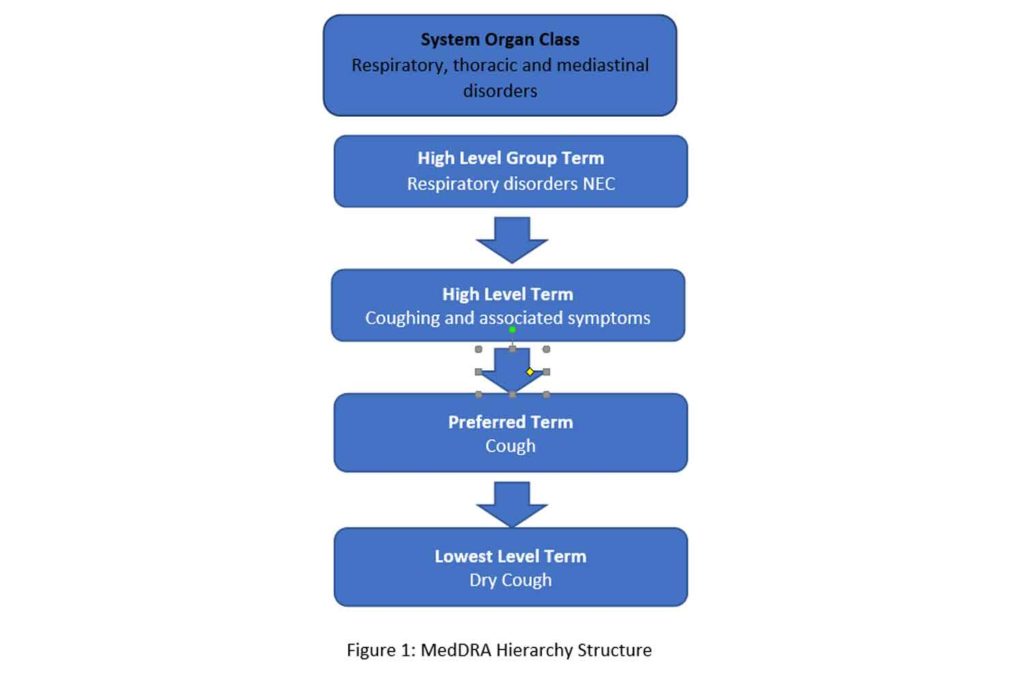
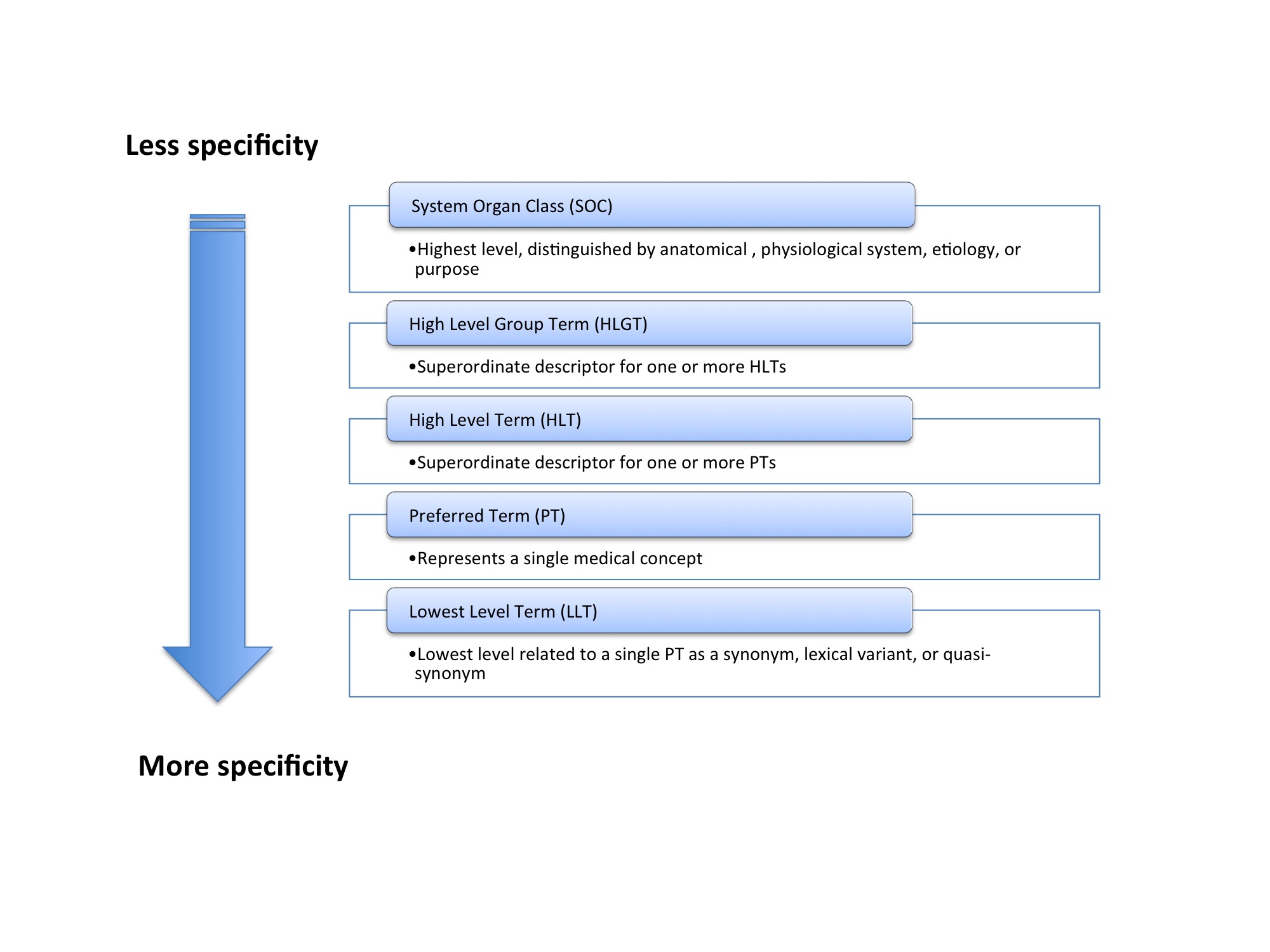
Table I from Public Assessment Report Decentralised Procedure Plenvu powder for oral solution (Macrogol 3350, sodium ascorbate, sodium sulfate anhydrous, ascorbic acid, sodium chloride, potassium chloride) Procedure No: UK/H/6370/001/DC UK Licence No:














.png)